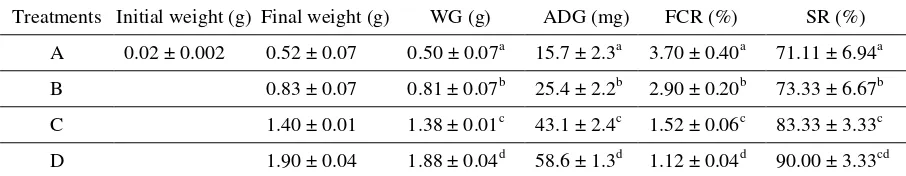
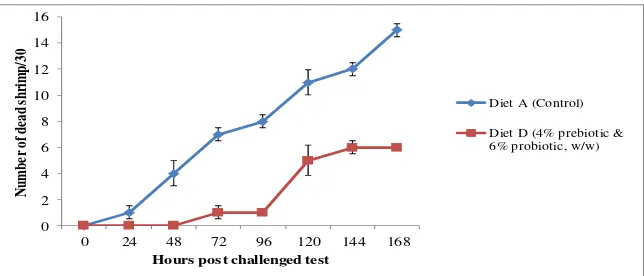
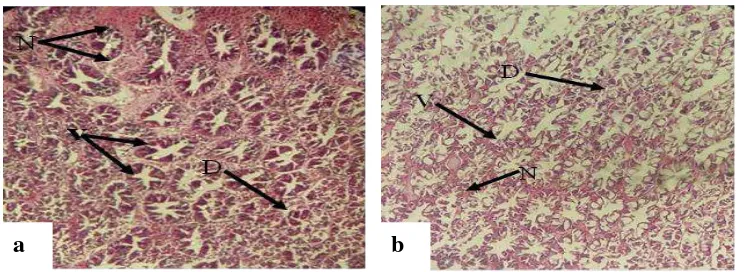
Harpeni et al., 2017 AI
Teks penuh
Gambar
Garis besar
Dokumen terkait
If SDK Manager doesn’t display its Android SDK Manager dialog box, you probably need to create a JAVA_HOME environment variable that points to your JDK’s home directory (for
Apabila menggunakan IPR untuk mengukur risiko likuiditas, maka pengaruh antara risiko likuiditas dengan IPR adalah negatif karena semakin tinggi IPR maka persentase
Dalam al-Qur’an membahas masalah ini yang mana uraian ini dimulai dengan sejarah Israil, beranjak kesejarah orang-orang Kristen dan akhirnya kepada orang-orang Islam. dinyatakan
Proses ini meliputi membuat campuran bahan yang seimbang, pemberian air yang cukup, pengaturan aerasi dan penambahan activator pengomposan.proses pengomposan dapat
Selanjutnya adalah diskusi kelompok ahli, dalam diskusi kelompok ahli mereka lebih efektif mendengar penjelasan dari siswa kategori tinggi dan mendengar pertanyan dari
Alih-alih adil secara gender, patriarki justru mengakibatkan ketimpangan relasi gender antara laki- laki dan perempuan yang dapat termnifestasi dalam berbagai bentuk
[r]
Undang-Undang Nomor 20 Tahun 1950 tentang Pemerintahan Jakarta Raya (Lembaran Negara Republik Indonesia Tahun 1950 Nomor 31, Tambahan Lembaran Negara Republik