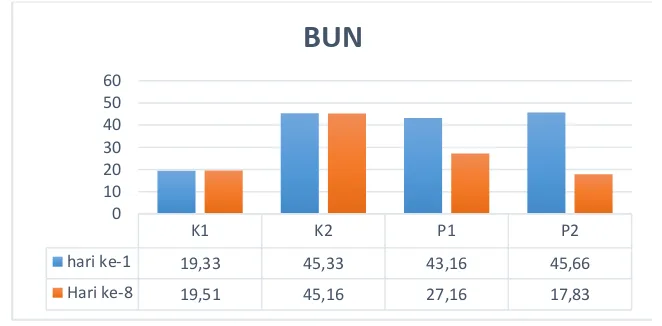
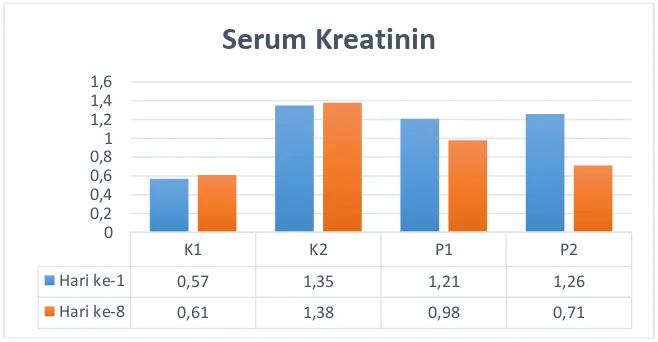
this PDF file The Effect of Avocado to the Profile of Blood Urea Nitrogen (BUN) and Creatinine in Rats (Rattus norvegicus)Induced with Meloxicam | Anshar | Jurnal Riset Veteriner Indonesia (Journal of The Indonesian Veterinary Research) 1 SM
Teks penuh
Gambar
Dokumen terkait
Untuk lokasi dengan demand terbatas atau kesulitas SITAC kabinet atau kondisi pencabangan dimungkinkan dilakukan design menggunakan ODP splitter dengan keluaran
Di dalam Kurikulum LPTK-PTK yang terstandar terdapat program profesi yang dipergunakan untuk uji kompetensi dan sertifikasi guru yang sudah ada di SMK dan bagi calon-calon guru
Isolasi mikroba adalah proses yang dilakukan bertujuan untuk memisahkan satu jenis mikroba dengan mikroba lainnya yang terdapat di alam dan menumbuhkannya dalam satu medium buatan,
TINGKAT SATUAN KERJA PERANGKAT DAERAH DI LINGKUNGAN PEMERINTAH KOTA DUMAI.. Satuan Kerja Perangkat Daerah
proses belajar mengajar, dari hasil evaluasi guru dapat mengetahui kekungan dan.. keterbatasannya dalam penyampaian materi dan untuk siswa, siswa dapat
Penambahan NaOH 17,5% pada sampel tandan kosong sawit yang. telah
Campur kode ke dalam ( innercode mixing ), yaitu campur kode yang diakibatkan oleh menyisipnya unsur-unsur bahasa lain yang bersumber dari bahasa asli (bahasa daerah)
Untuk mengetahui pengaruh pendekatan Realistic Mathematics Education (RME) terhadap hasil belajar siswa kelas VII SMP Islam Gandusari pada.