Role of male spatial distribution and co
Teks penuh
Gambar
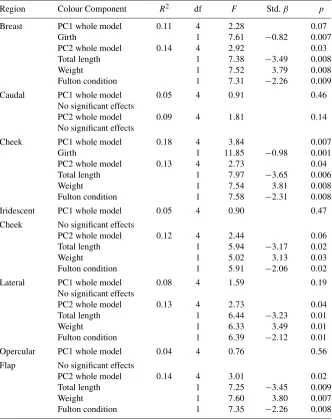
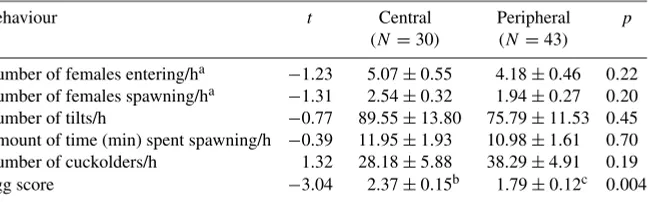
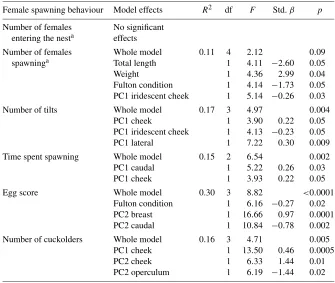
Garis besar
Dokumen terkait
Augusztinovics, M., The dynamics of retirement saving — theory and reality 111 Ayres, R.U., Structural Economics: Measuring Change in Technology, Lifestyles and the En 6 iron-..
[r]
Mendapatkan tanggapan peserta didik terhadap pembelajaran menggunakan multimedia pembelajaran interaktif game berbasis strategi scaffolding untuk meningkatkan
[r]
Sehubungan dengan telah dilakukannya evaluasi administrasi, evaluasi teknis, evaluasi harga untuk penawaran paket pekerjaan tersebut diatas, dengan ini kami sampaikan bahwa Pokja
These intermolecular forces depend on the molecular weight or the chain length, the presence of polar groups and their spacing and regular distribution along the
Manfaat dari penelitian ini adalah dengan adanya informasi ilmiah mengenai pengaruh budaya organisasional, lingkungan kerja, karakteristi individu terhadap motivasi kerja,
1) Dengan sikap tanggung jawab, responsif, dan santun siswa bersama guru menyimpulkan pembelajaran. 2) Bersama guru, siswamengidentifikasihambatan-hambatanyang dialami saat