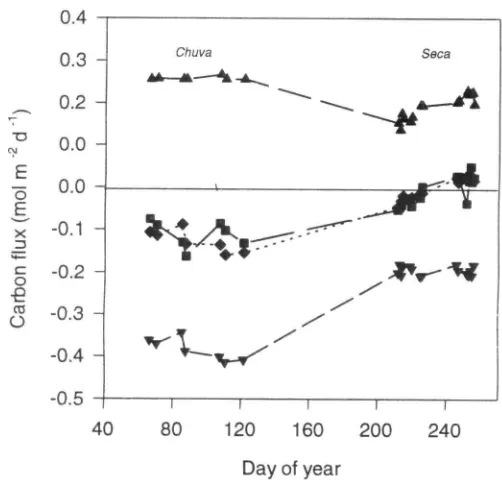
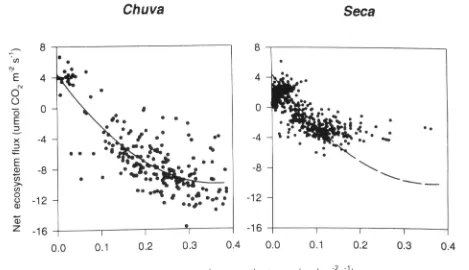
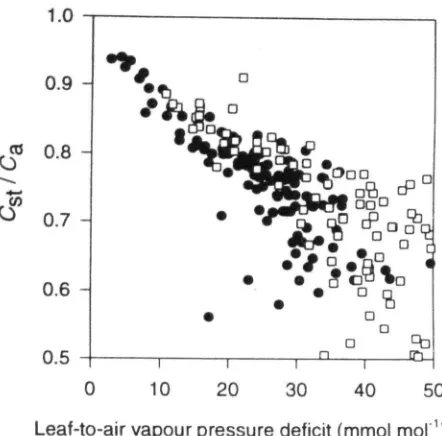
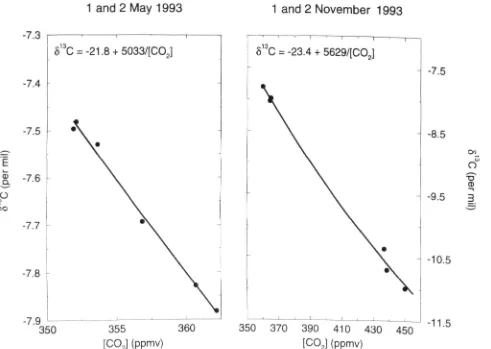
Fluxes of carbon water and energy over B
Teks penuh
Gambar
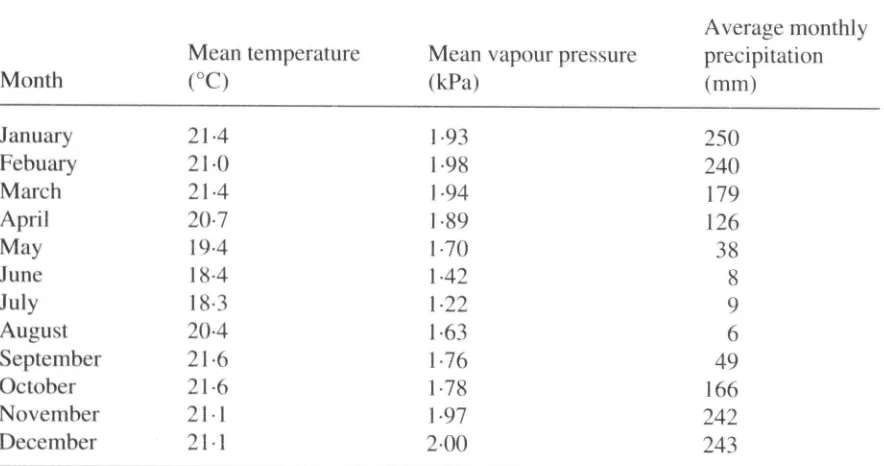
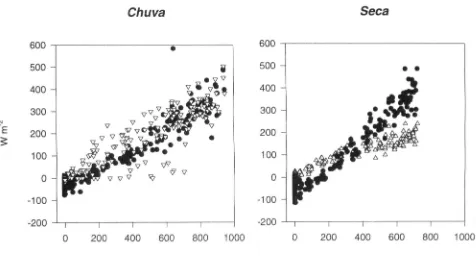
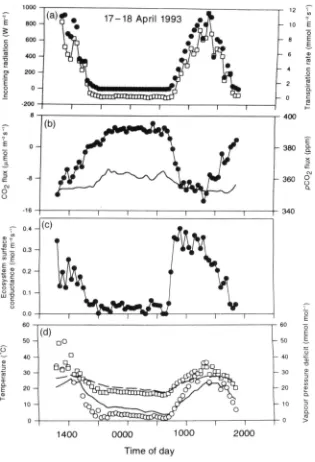
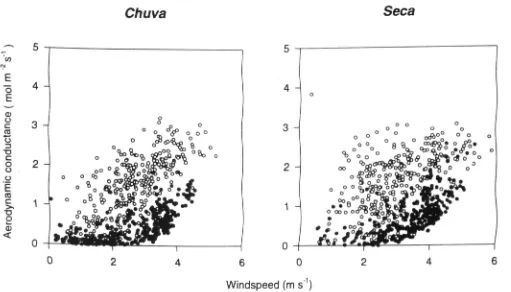
Garis besar
Dokumen terkait
Fase ini berupa berkontraksinya otot antar- tulang rusuk sehingga rongga dada mengembang. Pengembangan rongga dada menyebabkan volume paru-paru juga mengembang akibatnya tekanan
.lffi
digunakan peneliti untuk penelitian ini adalah member OLX.co.id yang pernah. melakukan jual beli pada situs
Sultan Agung adalah raja besar dari kerajaan……….. Tujuan Sultan Agung menyerang
Berdasarkan hasil evaluasi pengujian dengan time frame M15 sampai dengan H4 didapat bahwa indikator bekerja dengan baik di time frame H4 dengan menghasilkan total
Dokumen Kualifikasi Asli (BUKAN FOTOCOPY) serta sertifikasi keahlian tenaga ahli asli (BUKAN FOTOCOPY) yang disyaratkan di LDK pada Dokumen Kualifikasi. Demikian
RENCANA UMUM PENGADAAN BARANG JASA TAHUN ANGGARAN 2013. DAERAH SULAWESI TENGAH RESORT
Panitia Pengafaan Barang/Jasa pada Dinas Kesehatan Kabupaten Ponorogo akan melaksanakan Pelelangan Umum dengan pascakualifikasi untuk paket pekerjaan kontruksi sebagai berikut